
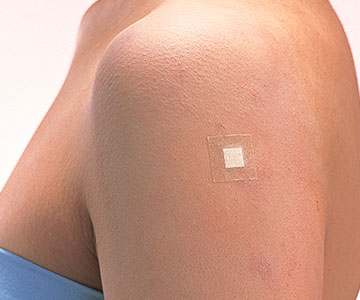
An adhesive bandage covering the puncture site. Hygienic application without touching the pad surface. The highly absorbent pad prevents blood from adhering to clothing.
Product information compliant with
EU Medical Device Regulation (EU MDR)![]()
Overview
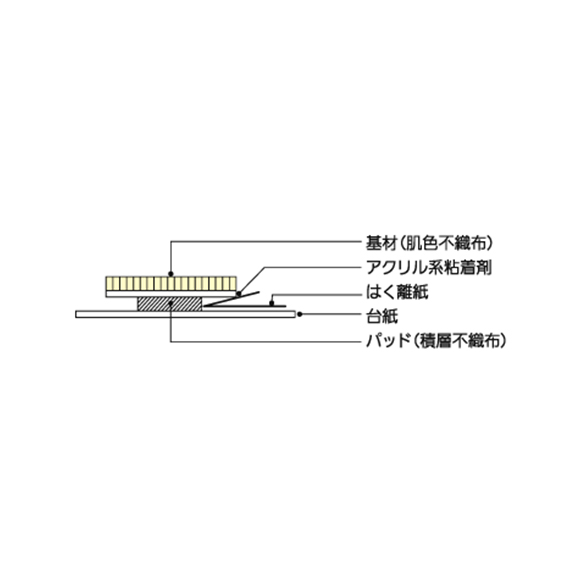
This is a bandage with pad for protecting the post-injection coating consisting of a pad of cellulose-based laminated non-woven fabric with high blood absorbency, a pulp / polyester-based non-woven fabric with high flexibility and air permeability, and an acrylic adhesive.
EOG sterilized
Usage
- Protective covering after injection, blood collection, infusion, or arthrocentesis
Features
Sanitary application without having to touch the pad and the adhesive side
- The turn up on the release liner allows the user to apply the tape sanitarily on the skin without touching the pad or adhesive surface, which is hygienic.
High absorbency (pad part)
- The pad of cellulose-based layered non-woven fabric with excellent absorbency gently protects the puncture area and prevents blood from getting on clothing.
Strong adhesion, fixing performance, low irritation (adhesive part)
- An acrylic adhesive that causes less skin irritation and gently conforms to the skin and offers excellent adhesion and fixing performance is used as the coating on the flexible, highly breathable pulp and polyester-based non-woven fabric of the tape.
Inconspicuous beige color
- The beige color of the tape blends in with the skin even when it is applied to exposed areas.
Sterilized low-count packaging to make it easier to use up the product
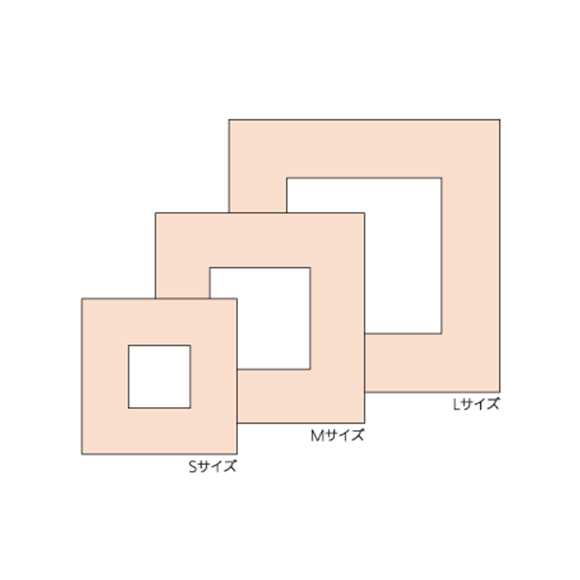
- Each package contains 10 S-size, 8 M-size and 6 L-size tapes in sterilized packs. The low product count makes it easier to use up.
Directions
-
1

Peel off the release liner (the folded upper and lower pieces, or only the upper piece) from the backing together with the tape.
-
2
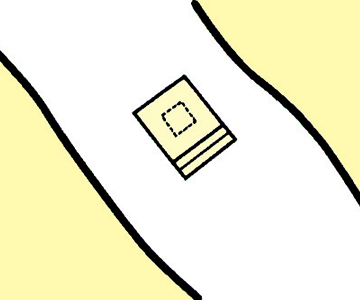
Place the pad over the punctured site and apply the adhesive surface, without the release liner, onto the skin.
-
3
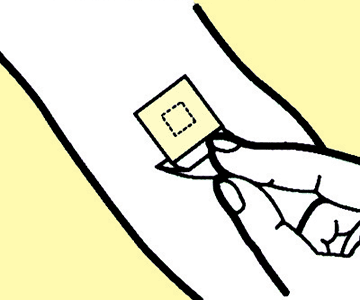
Pull the lower portion of the release liner (closer to the skin) away from the adhesive surface, and stick the exposed adhesive surface onto the skin.
-
4
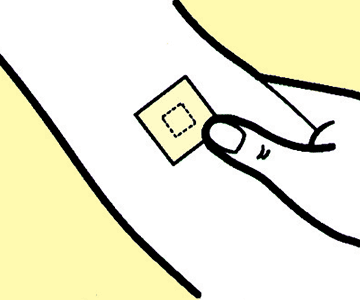
Press the tape area to attach it securely.
Precautions
Reuse prohibited
- If the wound becomes clinically infected while using the product, stop its use and conduct appropriate treatment.
- If any symptom of skin disorder (rash / redness, blisters, skin peeling, itchiness, etc.) appears while using the product, stop its use and conduct appropriate treatment.
- If during use the product gets wet or soiled, or if blood or exudate leaks from the pad, take it off and apply a new pad immediately.
- Do not use the product if the packaging is dirty, damaged or wet.
- Use the product immediately after breaking the seal.
- When removing the pad, peel it off slowly along the flow of the body hair to prevent skin damage.
Storage Instructions
- Store at room temperature, away from water, high temperatures, high humidity and direct sunlight.
Specifications
| Product number | Size | Per pack | Per box | |
|---|---|---|---|---|
| Adhesive area | Pad area | |||
| Small | 20mm x 20mm | 8mm x 8mm | 10 sheets | 1000 sheets |
| Medium | 27mm x 27mm | 13mm x 13mm | 8 sheets | 480 sheets |
| Large | 35mm x 35mm | 20mm x 20mm | 6 sheets | 480 sheets |
